
On the periodic table, the elements colored yellow, which generally border the stair-step line, are considered to be metalloids. Some of the groups have widely used common names, including the alkali metals (group 1. Elements that exhibit similar chemistry appear in vertical columns called groups (numbered 118 from left to right) the seven horizontal rows are called periods. Metalloids can also be called semimetals. The periodic table is an arrangement of the elements in order of increasing atomic number. The periodic table is a tabular array of the chemical elements organized by atomic number, from the element with the lowest atomic number, hydrogen. (b) The metallic atomic radius, rmet, is half the distance between the nuclei of two adjacent atoms in a pure solid metal, such as. Elements with similar properties are arranged in the same column (. Chemical elements are identified by the number of protons in the nuclei of their atoms, 1 known as the element's atomic number. (a) The covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as Cl 2. A table in which the chemical elements are arranged in order of increasing atomic number. The basic particle that constitutes a chemical element is the atom. Unfortunately, there was a slightly different system in place in Europe. A metalloid is an element that has properties that are intermediate between those of metals and nonmetals. A chemical element is a chemical substance that cannot be broken down into other substances by chemical reactions. 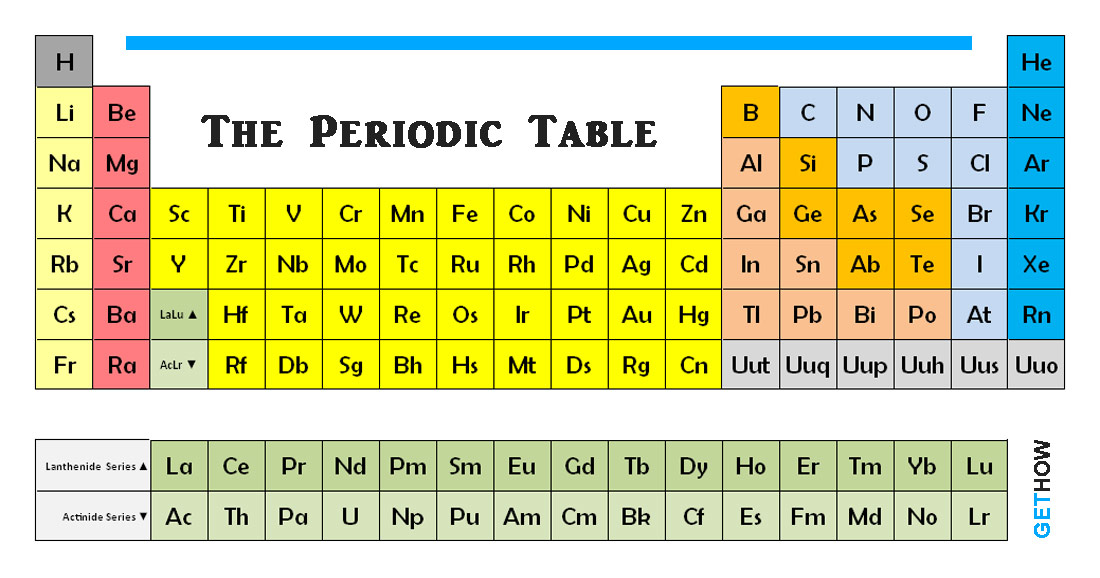
The first two groups are 1A and 2A, while the last six groups are 3A through 8A. The traditional system used in the United States involves the use of the letters A and B. There are two different numbering systems that are commonly used to designate groups, and you should be familiar with both. These two rows are pulled out in order to make the table itself fit more easily onto a single page.Ī group is a vertical column of the periodic table, based on the organization of the outer shell electrons. It alloys with copper, tin, and zinc to form amalgams, or liquid alloys. Mercury is silvery white and slowly tarnishes in moist air. Mercury is the only elemental metal that is liquid at room temperature. Atoms or ions with the same electronic configurations are said to be. Mercury, chemical element, liquid metal of Group 12 of the periodic table. Isoelectronic chemical species typically display similar chemical properties. The term means 'equal electric' or 'equal charge'. Periods 6 and 7 have 32 elements, because the two bottom rows that are separate from the rest of the table belong to those periods. Isoelectronic refers to two atoms, ions, or molecules that have the same electronic structure and the same number of valence electrons. Period 1 has only two elements (hydrogen and helium), while periods 2 and 3 have 8 elements. Not all elements form stable negative ions in which case the electron affinity is zero or even positive. So the more negative the electron affinity the more favourable the electron addition process is. 
A new period begins when a new principal energy level begins filling with electrons. The electron affinity is the potential energy change of the atom when an electron is added to a neutral gaseous atom to form a negative ion. There are seven periods in the periodic table, with each one beginning at the far left. The protons and neutrons account for almost all of the mass of an atom. the number of protons in the nucleus of an atom, which determines the chemical properties of an element and its place in the periodic table. There are 18 numbered groups in the periodic table the 14 f-block columns, between groups 2 and 3, are not numbered. In chemistry, a group (also known as a family) 1 is a column of elements in the periodic table of the chemical elements. \): Antimony.\) (Credit: User:Cepheus/Wikimedia Commons Source: (opens in new window) License: Public Domain)Ī period is a horizontal row of the periodic table. A period 1 element is one of the chemical elements in the first row (or period) of the periodic table of the chemical elements. In the periodic table of the elements, each column is a group.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
